Bæredygtighed
New Paths To More Efficient Hydrogen Production Discovered

Finding The Right Hydrogen Catalyst
Hydrogen could, in theory, be a perfect fuel to store energy and power applications that are hard to electrify. This is because it presents a few almost ideal characteristics:
- The byproduct of its combustion is only water
- And the same can be said when it is used to produce electricity in fuel cells.
- It can burn at very hot temperatures, making it a good alternative to natural gas in metallurgy, chemical processes, etc.
- It only requires water as a resource for its production.
- Hydrogen is itself non-toxic and non-polluting.
However, the rise of a hydrogen-based economy has been hampered by the difficulty of producing hydrogen in a cost-effective manner. This is due to the fact that most green hydrogen (produced from green energy) is made through electrolysis, a process for now mostly based on expensive catalysts like platinum, ruthenium, or iridium, each very rare and expensive metals.
So as long as no better production method for hydrogen exists, it is unlikely that we will manage to see it replacing fossil fuel at scale.
Luckily, this is quickly changing. We previously covered a few of these possibilities, notably turning plastic waste into hydrogen, using nanorods of nickel as an alternative catalyst, or using titanium and nickel scrap metals (swarf) produced during the manufacturing of metal parts. Some newer options are now being added by researchers.
The first is the creation of self-optimizing catalysts1 by researchers at the Johannes Gutenberg Universitat (Germany) and Technical University of Darmstadt (Germany), Max Planck Institute for Polymer Research (Germany), Harbin Institute of Technology (China), and Shandong University (China). It was published in Angewandte Chemie under the title “Self-optimizing Cobalt Tungsten Oxide Electrocatalysts toward Enhanced Oxygen Evolution in Alkaline Media.”
The second one is the invention of a method to turn sewage sludge into green hydrogen and animal feed2 by researchers at the Nanyang Technological University (Singapore), Monash University (Australia), and University of Hong Kong (China). It was published in Nature Water under the title “Solar-driven sewage sludge electroreforming coupled with biological funnelling to cogenerate green food and hydrogen”.
Fixing Hydrogen Catalysts
A recurring issue with all hydrogen-producing catalysts is that they degrade over time. This can be due to a deposit forming on the reactive metals, or the metal layer itself slowly degrading and losing components at each catalysis cycle.
This is especially problematic for expensive catalysts of the platinum metals group, but it is also an issue for other types of metal-based catalysts.
So, it is important that the German and Chinese researchers of the first study discussed here have observed self-optimizing behavior with their new catalysts.
“What’s so unique about our catalyst is that it actually enhances its performance over time, while conventional catalysts either maintain their performance at a consistent rate or even lose some of their performance because they are insufficiently durable,”
Dr. Dandan Gao – Research Lead at Johannes Gutenberg University Mainz
Cobalt-Tungsten Catalysis
Solving Hydrogen Production Chokepoint
The researchers focused on so-called 3d to 5d transition metal oxides, formulated as mixed metal.
These are able to perform a chemical reaction called oxygen evolution reaction (OER), which is half of the reaction occurring during electrolysis of water into hydrogen, in both of the most common electrolyzer designs (AEM and PEM electrolyzers).

Source: SpectroInlet
“There are two reactions during the splitting of water. The hydrogen evolution reaction (HER), which produces hydrogen gas, and the oxygen evolution reaction (OER), which produces oxygen gas. The OER represents the bottleneck for the whole reaction. That’s why we are so committed to developing a catalyst that can promote the OER half-reaction.”
Dr. Dandan Gao – Research Lead at Johannes Gutenberg University Mainz
However, these novel potential catalysts are still poorly understood, with little knowledge of what exactly happens at the atomic level during the reaction, or even the electrochemical form the metal takes.
This lack of understanding is a big hindrance to developing a commercially viable solution, as it also limits the ability to anchor the catalysts to a stable substrate.
One-Step Deposit Method
The researchers used a copper oxide (L−CuO) microflower substrate with a diameter of 3–5 μm, previously developed in their lab in 2020.
They then used a chemical deposit method to create a layer of cobalt-tungsten alloy at the surface of the copper substrate.
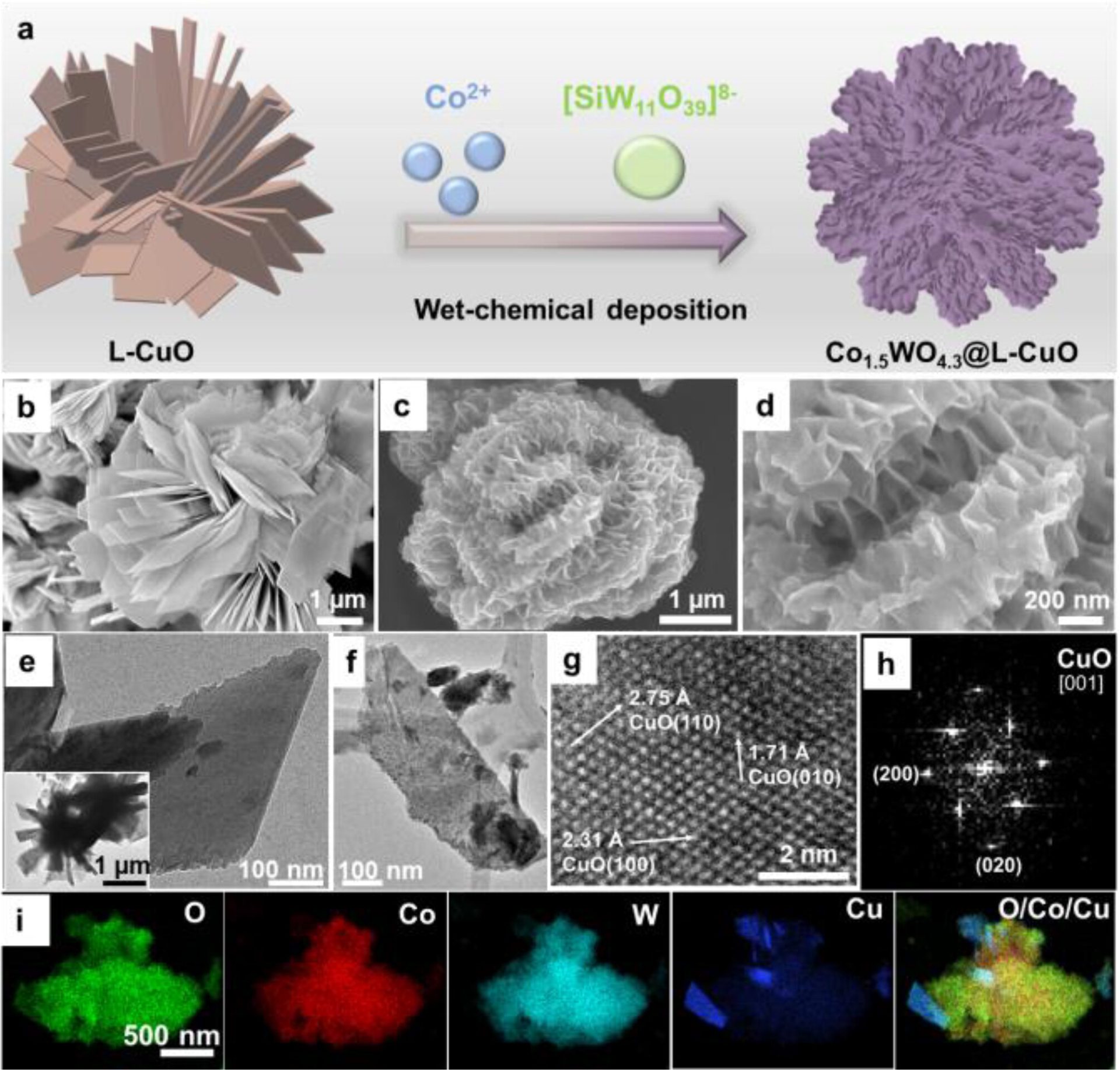
Source: Angewandte Chemie
Subsequent analyses revealed the complex microscopic structures of the material, using X-ray photoelectron spectroscopy (XPS), attenuated total reflection Fourier transform infrared (ATR-FTIR) spectroscopy, and Raman spectroscopy measurements.
It also confirmed that the catalyst is very firmly tied to the copper substrate.
“The development of viable and scalable deposition approaches is of utmost technological, economic, and ecological significance, enabling stable anchoring of OER pre-catalysts on selected promising substrates with high mechanical integrity”











