BioTech
The Biofoundry Revolution: Programming Life as a Factory

The Shift from Chemistry to Biological Factories
How Digital Code is Replacing Synthetic Chemicals
As soon as early scientists started to understand that the material world around them was made of separate, pure components, they started to work to understand it better. The early alchemist effort made way for enlightenment era scientists and the early modern period, when they discovered the separate elements and the basics of biology: cells, DNA, etc.
At the same time, the chemical industry was creating the early pharmaceutical industry, using synthetic chemical drugs to alter biological processes in patients, like using salicylic acid (aspirin) to lower fever.
Progressively, chemicals used in medicine and industry became increasingly more complex. However, in large part, the more complex a molecule, the more difficult it becomes to synthesize it by artificial chemical methods. And it becomes downright impossible for the most complex proteins or biochemical compounds.
Then, bioengineering allowed for the production by GMO microbes for the production of cheap and safe insulin, growth hormone, antibodies, etc, creating the field of biotechnology as a related, but separate field from pharmaceuticals.
This was a massive revolution in biochemistry and medicine, suddenly making abundant & cheap to produce compounds that were previously extremely expensive or impossible to obtain.
Today, many new technologies (big data, AI, automation, precise genetic engineering, advanced analytics, etc.) are converging to open another era of biosciences: the biofoundry revolution.
Beyond Nature: Redesigning Organisms for Industry
The biotech era was marked by the use of artificial genetic modifications to make microorganisms produce biomolecules of interest, usually medical products. This has proven extraordinarily profitable, as many of these molecules are either life-saving or high-value products that previously could only be harvested in small quantities from high-cost methods.
However, this has the inherent limitation of only being able to replicate what already exists in living organisms. But to this day, the production of plenty of materials and useful molecules is dependent on chemicals produced artificially, using toxic or carbon-emitting methods.
So while changing our energy system through EVs, batteries, and renewables is important, finding more green alternatives to chemical production is equally important for solving most of the modern world’s problems: plastic pollution, climate change, sustainable agriculture, non-polluting industrial production, biosecurity, incurable diseases, regenerative medicine, longevity treatments, etc.
And to all of these problems, a solution is now being deployed: the biofoundry model.
How the Biofoundry Model Works: A Tech Convergence
Multiomics, CRISPR, and the Rise of “Bio-Coding”
In recent years, understanding of biology and genetics has made tremendous progress. The key parts rely on a few novel technologies.
The first one is sequencing and genomics, which has become cheap enough to be done routinely for less than $1,000 per organism.
It is now combined with many other “-omics” (transcriptomics, proteomics, metabolomics, epigenomics, microbiomics, spatial biology) to create multiomics, a holistic understanding of all the multiple levels of complexity in living organisms.
Another novel technology is CRISPR, a new way of gene editing discovered in 2012, which has since become the most powerful way to edit the genes of all types of organisms, including for curing rare diseases.
Lastly, the emergence of big data, AI, and other forms of advanced analytics gave biologists the tools to process and make sense of the flood of data that multiomics has created.
When brought together, a whole new capability emerges.
The combination of tons of data from real biological multiomics with AI analysis means that the entire process to create complex molecules can be mapped, modeled, and even fully simulated in silico. This opens the opportunity for testing virtually thousands of possibilities or creating from scratch entirely new proteins with novel properties.
And thanks to CRISPR, putting these ideas into real microorganisms or plants has never been quicker, more precise, or easier, transforming them into well-controlled biological factories, or “biofoundries”, a subsection of synthetic biology.
As DNA is essentially a biological code, the ease of making GMOs and designing new biosystems turns biology very close to computer coding.
“Think of a cell. It’s kind of like a little machine that runs on digital code, very similar to a computer, except in this case the code—instead of zeros and ones, it’s A’s, T’s, C’s, and G’s. So synthetic biology is programming cells like we program computers, by changing the DNA code inside them. We’re sort of like cell programmers for hire. Our job is to make the cell do what our customers want.”
Jason Kelly – Ginkgo Bioworks CEO
From Plastic to Perfume: What Biofoundries Can Build
A lot of chemicals currently produced by the chemical industry could, in theory, be replaced by biologically made means. Either with the same molecule produced by living organisms, or with substitutes with similar properties.
For example, soil microorganisms and plants routinely produce in small amounts fertilizer, ethanol, or ethylene, all molecules currently mass-produced by the chemical industry. So, a higher yield or cheaper production by a living organism could have a much lower carbon impact.
Another target is reducing the dependency on fossil fuels by producing polymers, including textiles and plastics (1,4-Butanediol, 1,3-Propanediol, polyhydroxyalkanoates, polylactic, etc.) through biological metabolic paths.
High-value fragrances, amino acids, vitamins, silk, flavors like vanillin, and cosmetic ingredients like squalane or hyaluronic acid, all could also be mass-produced naturally at a cheap cost, at least in theory.
And of course, many newly invented biological molecules can form synthetic vaccines, anti-cancer treatments, alternative protein and food sources (cultivated meat, etc.).
Lastly, entirely novel products can be produced this way. For example, mushroom mycelium can create a viable alternative to leather and other textiles. Or carbon emissions can be directly recycled into useful products before ever reaching the atmosphere.
The Research-as-a-Service Business Model
Building Synergies
If the technology to do so has matured, it is, however, not so simple in practice to entirely rewrite the metabolism of a real living thing, while still keeping it productive as well.
This is why a growing trend is the outsourcing of this task to specialized companies that have the equipment, expertise, and the right biological material to pull it off. This “Research-as-a-Service” model, also sometimes called “organisms-on-demand”, allows for different projects and concepts to help each other across domains.
For example, a microorganism previously developed for the absorption of carbon emissions can also use that carbon to produce ethylene, a key precursor to countless chemical synthesis reactions. But a carbon credit-focused company would have no immediate use or experience with ethylene, while a chemical company might not have a carbon source handy. But by using the same biofoundry contractor, the two companies can develop synergies and make the process more efficient.
Similarly, a new optimized method for genetic modifications can be deployed for dozens of different applications, amortizing the cost of R&D on a wider range of projects.
Ginkgo Bioworks: The “DNA” of Synthetic Biology
No company has been more at the forefront of the “organisms-on-demand” than Ginkgo Bioworks. From its foundation in 2008 by five MIT scientists, the company was dedicated to producing GMO bacteria for industrial applications, with biotechnology, usually the focus of such activity, only a second thought.
Ginkgo was the first biotechnology company to join the famed Y Combinator start-up accelerator program in 2014. The company went public in 2021 through a SPAC merger and managed to secure the NYSE ticker DNA, previously held by biotech pioneer Genentech (before its acquisition by Roche).
Ginkgo Bioworks Holdings, Inc. (DNA -7.49%)
Since then, Gingko Bioworks has evolved into a key partners of many industrial, pharmaceutical, and agricultural corporations.

Source: Gingko Bioworks
For example, it developed new organisms for various research programs:
- Programmable microbes for gut diseases.
- Microplastics bioremediation.
- RNA therapeutics & vaccines.
- Recycling waste and contaminants.
- Controlling critical soybean diseases in Brazil.
- Replacing nitrogen fertilizers with bacteria
- Cannabinoids.
- Optimized manufacturing of biologics & peptides
- Production of active pharmaceutical ingredients (API)through scaled-up biocatalysis and fermentation.
- Molecular diagnostic solutions, through a proprietary enzyme database and expert enzyme designers.
- Cell therapy & gene editing.
Gingko’s Pivot: Selling Biosecurity for Autonomous Labs
Selling The Biosecurity Business
During the COVID pandemic, Gingko quickly expanded its biosecurity business, an activity that monitors biological risks, mostly for governments. It then evolved into an end-to-end bioradar platform.
That business provided invaluable benefits during the pandemic through our state and national testing programs and resulted in peak annual revenue over $300M. We were proud to help open over 5,000 schools nationwide.
However, this activity is relatively disconnected from the rest of Gingko Bioworks projects. So the company’s management has decided to divest it to a consortium of investors, forming a new standalone private entity named Tower Biosecurity, and Gingko will still retain a 20% equity stake.
From Service Provider to High-Value Partnerships
The “organism-on-demand” activity is for now the core of the company’s business, with the largest segment being food & agriculture and pharma & biotech. It has, however, declined in revenues between Q4 2024 and Q4 2025, due to a general decline in investment in biotechnology during this period.

Source: Gingko Bioworks
This segment has suffered from a relative uncertainty regarding its business model. Initially, Gingko planned to just provide the research capacity as a pure service, with a fixed price and a clear endpoint. This made Gingko extremely popular as a research partner.
However, this also meant that there were no residual royalties or extra revenues once the project was done, which meant Gingko was stuck on an endless treadmill of new projects, with technological prowess not really translating into revenues.
Since then, it has started to develop new organisms more with a partnership structure. For example, the delivery of a major milestone in a project with Merck, leading to a $9M payout in Q4 2024 and larger payments later in stage 2 of the project.
The need to restructure for more cash flow was taken seriously by the company from the other end of the equation, with strong effort made to reduce cash burn as well, down 73% in the past year. Meanwhile, the company also has no significant debt, reducing further financial risks.

Source: Gingko Bioworks
The Rise of the Modular, Autonomous Robot Lab
If cell engineering is the past center of the company, its future now lies in using AI and its own automated labs to reduce costs and improve the creation of new biofoundries.
Automated labs are a technology that Gingko has been working on for a while, as still today most of the work in biolabs is repetitive and tedious manual tasks, often consuming the bulk of the time of a workforce with Master’s degrees and PhDs.
To change this method, it has created an automated modular platform, able to perform without human intervention laboratory tasks like cultivating cells, transferring chemicals, microscopic analysis, etc.

Source: Gingko Bioworks
The most important characteristic of this design is its modularity. Each can then be connected with another in order to create a sort of “assembly chain” for scientific experiments and bioanalyses.
This solution is combined with a software offering, creating a flexible solution that can be adapted and modified in just days or hours, compared to more rigid research infrastructures requiring months of costly reconfiguration for new projects.

Source: Gingko Bioworks
This combination provides both the flexibility needed for research (compared to mass production), but also the automation needed to speed up research and reduce costs, as the automated lab can work more quickly than a human and 24/7.
Gingko is planning to offer this technology in two formats:
- “Build your own lab”, where it manufactures and services the automated lab modules, but the day-to-day operations and ownership belong to the client.
- Access Ginkgo’s own frontier autonomous lab via direct contracting of the service.
Datapoint: Generating 10,000 Experiments in Weeks
While the automation offering generates the biological data, Datapoint processes them into useful insights.
The key element is the quick generation of data that can guide further hypotheses, and the rapid iteration of new experiments to keep moving forward.
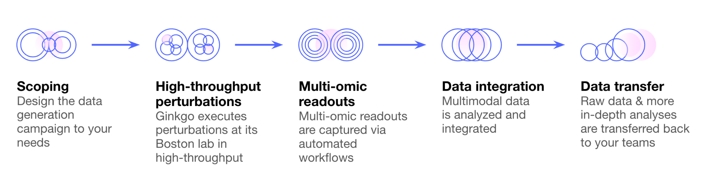
Source: Ginkgo Bioworks
With this service, Ginkgo can provide data that entirely belongs to the client, which is a competitive advantage in partnerships with other biotech or pharmaceutical companies.
Data can be generated in as little as 3 weeks, with over 10,000 in vitro chemical and genetic perturbations in each cell type, and a wide choice of analytical methods available to study the results.
The same system can be used for the quick generation of new antibodies, a type of molecule quickly becoming a key medicine in oncology and other medical fields. Ginkgo can screen up to 2,400 different antibodies in parallel, thanks to a $1B worth of automated wet lab infrastructure.
AI-Powered Bioresearch
Gingko established a partnership with OpenAI to use ChatGPT 5 in the context of bioresearch. It reported a drastic improvement in productivity.
“The company reports the system reduced cell-free protein synthesis reaction costs by 40% relative to state of the art, while running 36,000 experimental conditions across six iterative cycles.”
Human involvement was primarily limited to reagent preparation, loading and unloading, and system oversight, while experimental design, execution, data interpretation, and hypothesis generation were handled by the GPT-5-driven autonomous lab
The model used has been released as open source, and the AI-improved cell-free reaction mix can be ordered by the scientific community, turning Gingko into an important open source research tool for scientists worldwide.
Gingko was also awarded $47M by the US government for developing a large research facility for the Genesis Mission, a 32,000-square-foot site known as the Microbial Molecular Phenotyping Capability (M2PC). It will house over 100 automated analytical instruments and is expected to be fully operational for global researchers by 2030.
“The team will leverage advanced AI to decipher protein and pathway function, automate data generation and collection, and integrate DOE’s experimental and supercomputing systems with biotech and AI companies.”
The Long-Term Outlook: Is Ginkgo Finally Profitable?
From the creator of the biofoundry business model and an important actor in biosecurity, Gingko is now reinventing itself as a leader in automation of biological research and a key partner in AI-led R&D technology.
As biology meets AI, many industrial processes will progressively be replaced by greener bioprocess alternatives that are carbon-neutral, non-toxic, and cheaper. In that vision of the future, lifeforms are as programmable as computer code, but even more impactful on the real world.
This represents a massive opportunity for Gingko Bioworks, either for the project it is already working on, or for its automated lab design becoming a standard for most research teams in the long term.
Combined with an improvement to its cell engineering business model (with more royalties and more fair revenue-sharing contracts), this should help make Gingko profitable in the upcoming years.
(You can also read more about other synthetic biology companies in “Top 5 Synthetic Biology Public Companies”)











