BioTech
DNA Robots Explained: The Future of Medicine and Computing

Robot adoption is advancing rapidly, driven by declining costs, rising demand, and the integration of artificial intelligence (AI).
According to the World Robotics 2025 statistics on industrial robots, 542,000 robots were installed in 2024. This marked the “second-highest annual installation count of industrial robots in history – only 2% lower than the all-time high two years ago,” said Takayuki Ito, President of the International Federation of Robotics.
Besides factory floors, robots are also being actively deployed in airports, agricultural fields, offices, the military, and outer space as they evolve from rigid, pre-programmed machines into adaptive, intelligent systems.
Robots are no longer mere mechanical arms; rather, they are becoming smarter, smaller, and far more versatile thanks to advances in materials science, miniaturization, and AI.
This has resulted in a profound transformation in medicine, where surgical robots are enabling minimally invasive procedures with the kind of accuracy that hasn’t been achieved before. Microrobotics and bioengineered systems, meanwhile, promise targeted therapies that can significantly reduce side effects.
Even the idea of machines operating inside the human body is fast becoming a scientific reality.
Entering the Age of Molecular Robotics
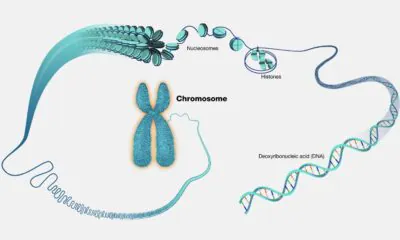
A new frontier in robotics is an emerging class of microscopic machines built from DNA and designed to operate inside the human body.
The concept of DNA nanotechnology was first introduced several decades ago by Professor Nadrian Seeman, widely recognized as the field’s founding father. The progress in the field, however, has been slow in the early years due to high costs and the immaturity of DNA synthesis technologies.
Advances in DNA chemical synthesis in the early 21st century lowered production costs drastically and accelerated the development of DNA nanotechnology.
A major breakthrough actually occurred two decades ago, when Paul Rothemund introduced the DNA origami technique, which enabled the creation of nanostructures through the bottom–up DNA self-assembly and has since become one of the most widely adopted strategies for constructing DNA robots.

These tiny devices are built from biological molecules that can navigate the body, interact with cells, and perform highly specific tasks. Given that these systems are built from the same fundamental material that runs through the body, unlike traditional robots, they can operate seamlessly within biological environments.
A new study published in the journal SmartBot1 highlights just how far this field has come. It demonstrates the growth of DNA machines from early ideas to more complex, practical, and capable systems that could one day deliver drugs directly to diseased cells or even identify and neutralize viruses inside the body.
Their potential extends much further, encompassing single-molecule analysis, atomic-level nanofabrication, and even building extremely small computing devices and data storage systems.
In their detailed review, researchers from Peking University (PKU) explain how DNA is being used to create functional machines. The very same DNA, deoxyribonucleic acid, that carries genetic information in almost all living organisms, is being used here. It is an ideal and versatile building material for constructing microscopic robots with complex geometries, precisely defined dimensions, and multifunctional capabilities.
That is because of DNA’s ease of synthesis, its capacity for precise self-assembly, its structural stability, and its programmability. The molecule offers a particularly unique advantage in “mechanical programmability”, noted the study. While single strands (ssDNA) provide flexibility, double-stranded sections (dsDNA) add structure to designs, and together they give a clear design toolkit.
Due to these properties, along with advances in structural DNA nanotechnology, DNA robots, often referred to as DNA nanomachines and nanorobots, have been rapidly evolving.
To create these tiny robots, scientists are combining traditional robotics with DNA folding techniques, enabling movement and reliable task performance with high accuracy.
DNA robots are still in early stages, though, and face significant barriers. Despite the challenges, the field is advancing as scientists learn to design DNA structures that can bend, grab, fold, and move on command.
With that, the work underscores a future in which these programmable biological machines can serve as precision tools for diagnosis, treatment, and disease prevention, potentially transforming medicine at its core.
“The robots of tomorrow won’t just be made of metal and plastic,” noted the research team. “They will be biological, programmable, and intelligent. They will be the tools that allow us to finally master the molecular world.”
Cracking the Challenge of Molecular Motion
To build molecular machines, researchers have been exploring DNA for a long time, exploring how it can be engineered into working machines.
The designs of early DNA devices were very simple; they could open and close or move along a track. While simple, they proved that motion at the molecular level was possible.
Now, scientists are going further with creative design approaches, including incorporating flexible components, constructing firm DNA joints for stability, and using origami-inspired folding methods.
In DNA origami, long strands are folded into complex shapes. Researchers use hundreds of smaller strands to guide a single one into detailed shapes such as boxes, cages, and gears. While some designs can have thousands of components, others can act as tiny switches, walkers, or grippers.
So, researchers are applying principles from traditional, large-scale robotics at the nanoscale, enabling DNA-based systems to perform repeatable, controlled tasks.
But turning DNA into machines requires not just structure but also motion, and the extremely small size of these DNA robots poses a major challenge for guiding their motion in the chaotic, constantly changing molecular environment.
| Key Area | Current Situation | System Focus | Why It Matters |
|---|---|---|---|
| Core Material | Conventional robots rely on metals, chips, and motors. | Use DNA as a programmable construction material. | Enables machines to function inside biological environments |
| Structural Design | Mechanical systems are assembled from rigid components. | Fold DNA strands into boxes, joints, and cages. | Allows precise nanoscale architectures to be built |
| Motion Control | Random molecular motion disrupts nanoscale machine behavior. | Guide movement using DNA reactions or signals. | Makes predictable molecular action possible |
| Medical Use | Many therapies still affect healthy tissue as well. | Deliver drugs only at diseased cellular targets. | Could improve precision while reducing side effects |
| Manufacturing Scale | Producing identical DNA machines remains difficult and costly. | Develop reliable, high-yield bio-manufacturing methods. | Essential for real-world deployment beyond laboratories |
| Future Development | Design tools and simulations remain underdeveloped today. | Use AI to optimize design and behavior. | Could accelerate progress across medicine and computing |
To control how these machines move, scientists have developed systems that enable these machines to behave in predictable ways. This includes biochemical reactions and physical signals such as heat, light, magnetic fields, and electric fields.
When it comes to biochemical control, the method researchers are using is DNA strand displacement, a process that allows precise programming of movement with the help of “fuel” and “structure” DNA sequences. Here, one strand kicks another out of position, acting like a molecular switch that can trigger a set movement.
However, every method carries trade-offs, requiring scientists to balance precision against speed.
For instance, chemical control provides accuracy and versatility but produces waste molecules and requires extensive experimental screening. Meanwhile, external physical signals act quickly, but they affect surrounding systems. They move whole structures but struggle to enable independent joint-level control.
It is by combining these strategies that scientists provide a toolkit to fine-tune the behavior of DNA machines with great precision. When it comes to the application of these microscopic machines, the study notes that they go far beyond the laboratory.
For starters, DNA robots can be of immense help in precision medicine, where they can act as “nano-surgeons” inside the body, identifying diseased cells and delivering therapies to those cells.
In a DNA robot example, SARS-CoV-2 was captured from saliva within half an hour using four flexible fingers, and it performed as well as conventional lab tests. In another case, the robot carried a clotting drug to tumor blood vessels in mice and delivered it only once it had reached the target, showcasing its potential as an autonomous drug-delivery system.
DNA robots can also serve as programmable templates for arranging materials, thereby enabling molecular optical devices, computing devices, and ultra-dense data storage systems more efficient than current technology.
DNA guides, nanoparticles, and light sources have already been arranged into ordered patterns. In related experiments, researchers have also printed chemical marks onto synthetic DNA and encoded images without writing every base afresh. So, the possibilities with these DNA machines are simply outstanding.
But of course, this is all still in early experimental stages. As they remain far from practical, real-world applications, these DNA robots are best understood as proof of concept. In fact, actualizing these machines faces several challenges. Scale is one of the issues.
When we move from large-scale systems to nanoscale (∼100 nm, about 1/500 to 1/1000 the width of a human hair), the precise control of these machines becomes difficult due to Brownian motion, which is the small, random motion of nanoparticles, and thermal fluctuations. The study noted:
“Although macroscopic robotics offers valuable conceptual and analytical frameworks, translating its principles to the molecular- and nano-scale demands a profound redefinition of mechanical design and motion control under stochastic, thermodynamic, and biochemical constraints.”
That’s why many existing DNA robot designs are simple and operate in isolation. Their usefulness in complex real-world environments is also limited.
But future systems need to be scalable, reconfigurable, and functionally integrated, which depends on adopting advanced modularity and translating macroscale mechanical principles to the molecular level.
Then there’s the matter of knowledge gaps. Even today, researchers lack detailed information about and understanding of the mechanical properties of DNA structures. Computational modeling and simulation tools for predicting how these structures behave at such microscopic scales are not yet fully developed.
Manufacturing presents another obstacle. Producing identical DNA machines at scale is necessary for their real-world applications, but it requires cost-effective, high-yield, and reliable methods that remain difficult to achieve.
Overcoming all these barriers, the study says, requires collaboration across disciplines: mechanical engineering, computer science, medicine, chemistry, and biology.
More specifically, scientists propose solutions like advancing bio-manufacturing methods, creating standardized DNA “parts libraries,” and using AI to improve design and simulation.
Deep learning and LLMs, as per the study, present “transformative opportunities for advancing the design and analysis of DNA machines” as well as simulation and dynamics analysis. The technology can uncover structural patterns from large datasets, predict folding pathways, optimize sequence configurations, and automate design evaluation, significantly accelerating the innovation cycle.
Progress in these areas will help scale DNA robots and integrate them into practical applications in science, healthcare, manufacturing, and beyond.
Investing in DNA Robotics Tech
In the world of medical robotics, Illumina, Inc. (ILMN ) stands out for its core expertise in DNA technologies and strong positioning in genomics-driven medicine. While the company isn’t building DNA robots itself, it is a big enabler of the entire ecosystem that makes such innovations possible.
A global leader in DNA sequencing, the company provides the foundational tools that enable research into DNA-based systems, including DNA nanotechnology and robotics. It is also deeply embedded in the shift toward personalized and molecular medicine.
The company’s products are used in research and clinical as well as oncology, life sciences, reproductive health, agriculture, and other segments. As for its customers, they include academic institutions, genomic research centers, hospitals, government laboratories, commercial molecular diagnostic laboratories, biotechnology, pharmaceutical, and consumer genomics companies.
Illumina’s goal is to improve human health by unlocking the power of the genome. Just last month, Illumina announced a strategic collaboration with Veritas Genetics to bring whole-genome sequencing into everyday healthcare through insurance systems.
This collaboration supports an integrated data ecosystem to advance research, drug discovery, and clinical trial optimization. More importantly, it marks a shift from treating diseases to predicting and preventing them using genetic data.
“Genomics is increasingly moving upstream in healthcare, from diagnosing disease to helping prevent it,” said Rami Mehio, general manager, BioInsight at Illumina. “By combining Illumina’s sequencing and informatics backbone with Veritas’ patient-ready reporting, this collaboration represents an important step forward in making preventive genomics actionable, accessible, and integrated into everyday healthcare.”
A couple of months before that, Illumina introduced the Billion Cell Atlas, the world’s largest genome-wide genetic perturbation dataset, which could make DNA robots practical and programmable.
This massive dataset maps how billions of cells respond to genetic changes, built using CRISPR and sequencing. The first tranche of the company’s program to build a 5 billion-cell atlas over three years, which will make it “the most comprehensive map of human disease biology to date,” is designed to train AI models and accelerate drug discovery in partnership with Merck, AstraZeneca, and Eli Lilly and Company.
“We believe the cell atlas is a key development that will enable us to significantly scale AI for drug discovery,” said Illumina CEO Jacob Thaysen. “We are building an unparalleled resource for training the next generation of AI models for precision medicine and drug target identification, ultimately helping map the biological pathways behind some of the world’s most devastating diseases.”
(ILMN )
Amidst these advancements, Illumina’s shares, with a $19.5 bln market cap, are trading at $127.74, up 74% in the past year. Its EPS (TTM) is 5.48, and the P/E (TTM) is 23.32.
As for Illumina’s financial strength, for Q4 of 2025, the company reported $1.16 billion in revenue, a 5% increase from 4Q24. Its GAAP operating margin was 17.4%, and its non-GAAP operating margin was 23.7% while GAAP diluted EPS came in at $2.16 and non-GAAP diluted EPS was $1.35.
During this period, capital expenditures were $54 million while cash flow from operations was $321 million. At the end of the year, the company had $1.63 billion in cash, cash equivalents, and short-term investments.
For the full fiscal year 2025, Illumina posted a revenue of $4.34 billion. Meanwhile, its GAAP operating margin was 18.6%, its non-GAAP operating margin was 23.1%, its GAAP diluted EPS was $5.45, and its non-GAAP diluted EPS was $4.84.
Last year’s capital expenditures amounted to $148 million, while cash flow from operations was $1.1 billion, and free cash flow was $931 million.
Talking about the “strong finish to 2025,” Thaysen said this marks “a return to growth through disciplined execution against our strategy,” with momentum built in the second half of the last year, especially with the increasing adoption of NGS-based testing in clinical markets.
Notably, Illumina made progress in China, with an export ban on its sequencers lifted. But it remains on the Unreliable Entities List (UEL), requiring approvals for instrument purchases.
For the current year, Illumina expects a 4% to 6% increase in revenue to $4.5 billion and $4.6 billion. The growth includes 1.5% to 2% benefit from the recently closed SomaLogic acquisition, which expands the company’s multiomics portfolio and strengthens its position in NGS-enabled proteomics.
Latest Illumina, Inc. (ILMN) Stock News and Developments
Conclusion
Robots have been redefining just what machines can achieve. They have been enhancing productivity, safety, and discovery across several domains. From industrial automation to planetary exploration, the continued evolution of robots highlights a broader trend toward more capable systems being integrated more deeply into our lives.
In medicine, the emergence of biologically compatible robots like DNA-based systems is enabling unprecedented precision in drug delivery and viral targeting.
More importantly, these systems promise not only more precise treatments and improved patient outcomes but also a new way to study processes at the molecular level and to build smaller, more powerful devices through DNA-guided assembly.
While significant challenges in scalability, stability, and long-term safety must be addressed before these technologies can move from laboratory research to clinical practice, the potential benefits are significant. And as robotics continues to shrink in size and expand in capability, it can provide a future where medicine is intelligently executed from within.
Click here to learn if AI can rewrite our DNA.
References
1. Xu, N., Zhang, X., Liu, Y., Wang, C., Li, J., Chen, Z., Zhao, H., Sun, K., Zhou, Q., Yang, F., Wu, T., Guo, S., Li, Y., Huang, J., Deng, D. & Bao, X. Designer DNA-based machines. SmartBot (2026). https://doi.org/10.1002/smb2.70029